DOUBLE DONOR CHIMERISM DETECTION, QUANTIFICATION AND MONITORING
Increasingly, the double donor technique for hematopoietic stem cell transplant is replacing the single donor approach. In most cases, physicians use the double donor approach because it improves the engraftment of the stem cells in the recipient. Usually, this is accomplished with an initial transplant of bone marrow along with either cord blood or peripheral blood stem cells. There is also the potential for the initial transplant to fail, thus, a second donor will decrease the chances of failed engraftment or disease relapse. In either case, the recipient has a chimera of three different genotypes. Theoretically, one of the donors and the recipient stem cells will begin to disappear, while the other donor cells engraft disappear within the host.
ChimeRMarker® software solves the technical challenges for chimerism monitoring of a double donor transplant by accurately identifying peak origins and quantifying the amount of Donor/Recipient chimerism with many shared alleles (Figure 1). ChimeRMarker software tackles these technical challenges by applying a Chimertyping panel to each sample for automatic peak identification (Figure 1) and accurate size calling algorithms, quantifying the amount of donor/recipient chimerism in one step.
Double Donor Case Report with Donor, Recipient and Shared peak notation
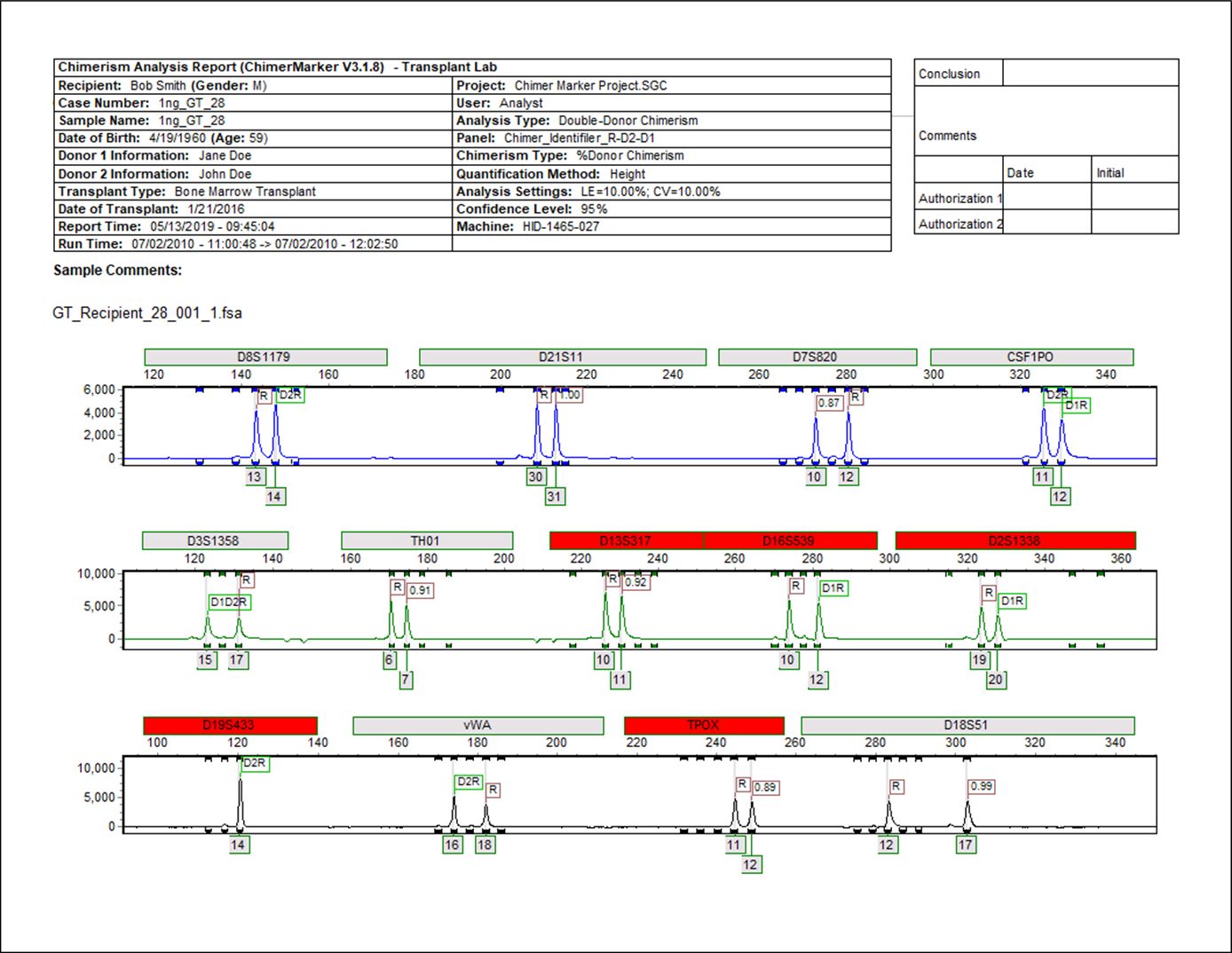
Figure 1: Each allele in the Post-BMT sample is automatically identified and displayed in ChimeRMarker software. Loci labeled in red were selected by the user to be ignored in the calculations or recognized by the software to be uninformative. The header provides electronic verification of date and time, user electronic identification and analysis parameters for complete record keeping.
Double Donor Case Report Chimerism Calculation Results and Statistics
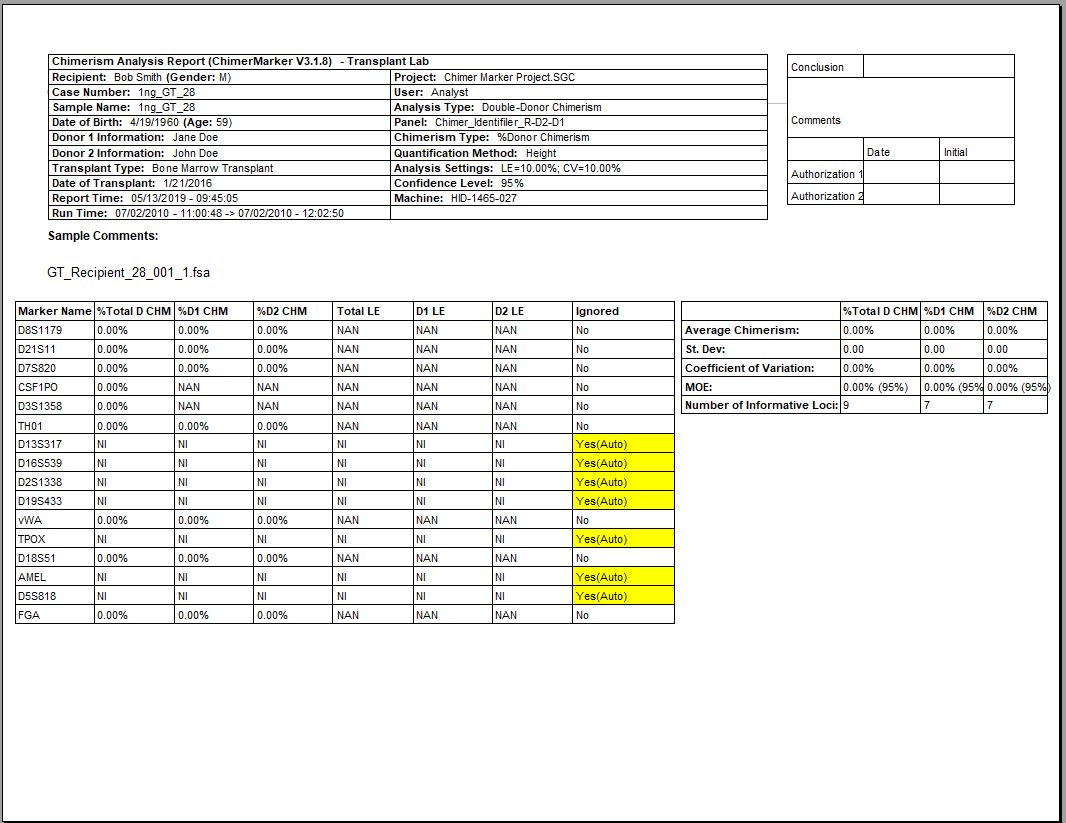
Figure 2: The second page of each double donor report displays deconvolution of Donor 1 and Donor 2 contributions along with a total Donor chimerism. ChimeRMarker software has the ability to quantify % donor or % recipient, and display the results in the summary final report.
Webinars:













