

ARMS™ (Amplification Refractory Mutation System, Newton et al. 1989) allele specific amplification technology, detects point mutations, insertions or deletions in DNA. The ARMs technology is applied to cystic fibrosis research in chemistries such as GenProbe® EU-2 (Elucigene). The Oligonucleotide-Ligation-Assay (OLA) is used to detect mutant point mutations in cystic fibrosis research in chemistries such as the Abbott® Cystic Fibrosis Genotyping Assay.
GeneMarker genotyping software is compatible with the data files from all major CE genetic analyzers, custom and commercial chemistries and Windows® 7-10 OS. GeneMarker software has embedded applications to streamline analysis of ARMs and OLA generated data for cystic fibrosis research.
See figures 1 and 2 for an overview of linked ARMs application which compares file pairs for a given individual and presents results in a concise clinical research report. GeneMarker software is ideal for analysis of samples from GenProbe® EU-2 for cystic fibrosis.
See figures 3 and 4 for an overview of the analysis results of OLA data from Abbott® Cystic Fibrosis Genotyping Assay. The main analysis screen provides rapid visual evaluation of mutant alleles in the electropherogram allele report. Mutant allele labels in electropherogram are purple (compared to gray normal allele labels) for instant visualization of mutant alleles.
Analysis of GeneProbe EU-2 Cystic Fibrosis Data
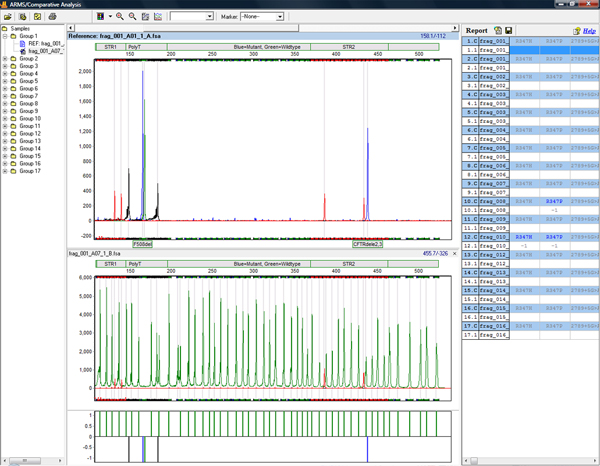
Figure 1: Review Results from GeneProbe EU-2 in the ARMs application. The application includes automated grouping of two dye chemistry, trace comparison of mutant and wildtype allele calls, a difference histogram and report table.ARMS/Comparitive Analysis Final Report.
Clinical Research Report
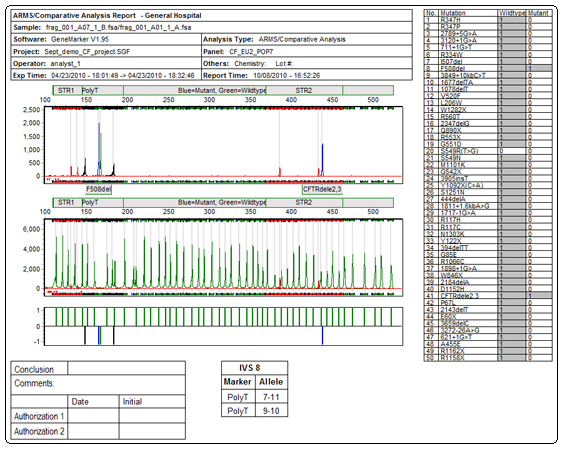
Figure 2: Final Report includes Customized Header, Electropherograms, Difference Histogram, Report Tables and Authorization/Conclusion box.
Abbott Cystic Fibrosis Genotyping Assay Results

Figure 3: Mutant alleles are labeled in purple.
Abbott Cystic Fibrosis Genotyping Assay Allele Report
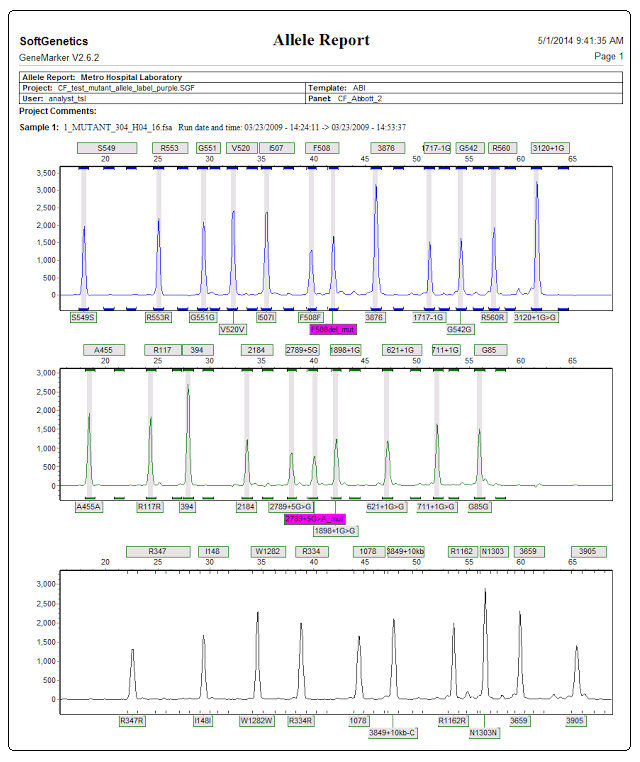
Figure 4: The Abbott CF allele report displays heterozygous mutations and labels them in purple.
MLPA® is the registered trademark of MRC Holland